Our new Annals of Internal Medicine paper, Efficacy of BNT162b2 Bivalent Vaccine Against Delta and Omicron Variants in Pediatrics, is online on January 9, 2024! Senior authors are Yong Chen, Christopher B. Forrest, and Jeffrey S Morris, and Lead authors are Qiong Wu and Jiayi (Jessie) Tong.
Penn Medicine’s news on our study: https://www.pennmedicine.org/news/news-releases/2024/january/analysis-covid-vaccine-strongly-effective-in-young-people.
Check out the paper: [https://www.acpjournals.org/doi/full/10.7326/M23-1754?journalCode=aim]
Our key findings are:
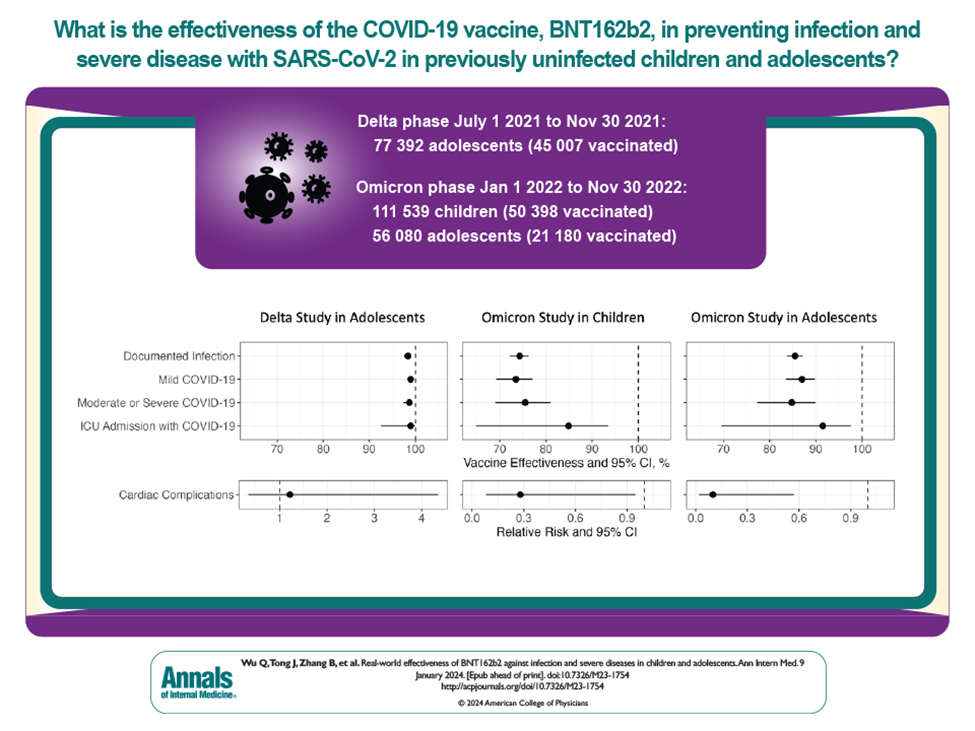
- During the period when the Delta variant was dominant, vaccinated adolescents were found to be approximately 98% less likely to be infected than those who did not receive it. During the Omicron wave, vaccinated children were 74% less likely to be infected than their unvaccinated peers; this protection was estimated as 86% among adolescents.
- The vaccine’s protection against severe illness and ICU admission was similarly high in all cohorts.
- No indication of increased cardiac risks was found during either Delta or Omicron phase for the vaccinated group.
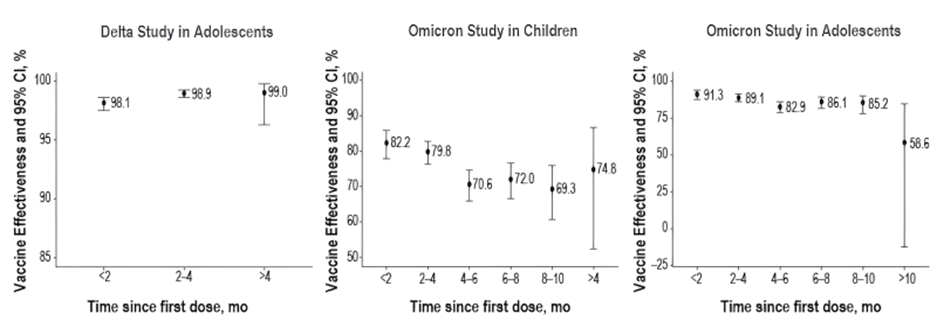
- When the Delta variant was dominant, there was no evidence of significant waning in vaccine’s effectiveness over the subsequent 4 months. During the Omicron wave, the data showed some reduction in effectiveness near the end of 4 months following vaccination.
Our study involves large USA pediatric cohorts totaling 250,000 children and adolescents from PEDSnet, a national network of U.S. pediatric health systems. The study covered a diverse and representative pediatric population from primary care, specialty care, emergency department (ED), testing centers, and inpatient settings. By using electronic health records data from PEDSnet, we defined a rich set of variables for the analysis, including vaccination status, COVID-19 outcomes (documented infection, severe illness, and ICU admission), cardiac complications, healthcare utilization (i.e., negative COVID-19 tests), pediatric comorbidity indicator, and preexisting chronic conditions.
A key issue in the electronic health records data is the under-reported vaccination status that can lead to biased vaccine effectiveness estimates. Children or adolescents could get vaccinated from various places, whereas only some of these places share the vaccination data with the health system. Consequently, the “vaccinated” status in the data was likely to be true, whereas the “unvaccinated” status in the data could be truly vaccinated.
To address the potential bias from the under-reporting issue, we developed and employed a novel comparative effectiveness research method. Our study was the first to rigorously address the under-reporting issue in vaccination status by health systems in the USA.
In addition to adjusting for a rich set of confounding variables and under-reported vaccination status in our proposed method, we performed negative control experiments to further mitigate potential residual bias. We pre-specified 40 negative control outcomes that are not causally related to the vaccines. By applying the same method to each of these outcomes, we estimated the distribution of the potential residual biases, which was used to adjust our primary results.